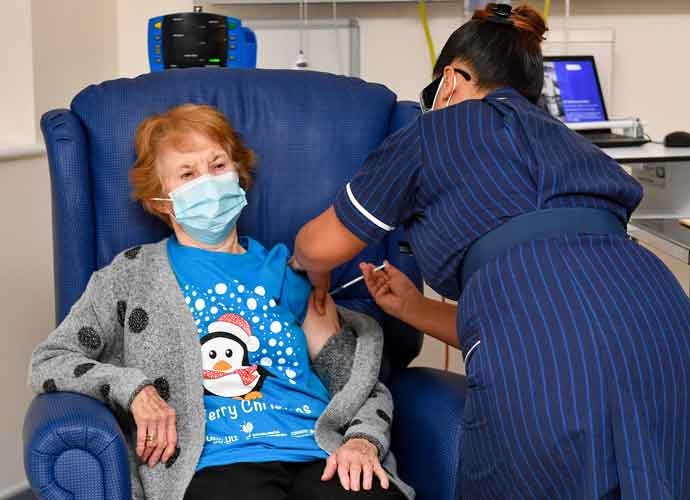
FDA Expected To Authorize Pfizer COVID-19 Vaccine For Adolescents
The Food and Drug Administration is expected to authorize use of the Pfizer-BioNTech coronavirus vaccine for adolescents 12-15 years old) in the next few days.
Pfizer recently announced that its vaccine is at least as effective in adolescents as in adults, according to an adolescent vaccination trial conducted by the pharmaceutical company.
The CDC convened an advisory panel to review Pfizer’s clinical trial data, as well as amend the emergency authorization use for the vaccine.
According to Pfizer’s clinical trial data, none of the adolescents who received the vaccine contracted symptomatic infections. Additionally, Pfizer reported that the trial volunteers produced strong antibody responses. Overall, the data seems to match up with vaccination data for people in the age group of 16 to 25.
Subscribe to our free weekly newsletter!
A week of political news in your in-box.
We find the news you need to know, so you don't have to.
Some doctors are wary about administering a vaccine to a group that is at a low risk of contracting severe COVID, arguing that it would be better used for adults in countries with vaccination rates.
President Joe Biden is under pressure to give away the country’s vaccine supply to other countries in need. The U.S. vaccine supply is fairly substantial, especially when compared to other countries: the U.S. has 31 million doses of Pfizer, 25 million doses of Moderna, and 10 million doses of Johnson & Johnson which have been delivered but not yet administered.
Indeed, just last week, the White House announced its plans to send 60 million doses of AstraZeneca to other countries, despite the vaccine not yet receiving authorization from American regulators.
Moderna will soon receive the results from its own clinical trial on adolescents ages 12 to 17. Modern will also receive results from a trial on children 6 months to 12 years old later this year.
This updated plan would allow millions of Americans to get vaccinated in the coming weeks, which would, in turn, raise the country’s level of immunity and reduce hospitalizations and deaths.
Even though over 100 million U.S. adults have been fully vaccinated, 44 percent of adults have not yet received even one dose.
Get the most-revealing celebrity conversations with the uInterview podcast!








Leave a comment