FDA Advisory Committee Votes To Recommend Novavax Covid-19 Vaccine
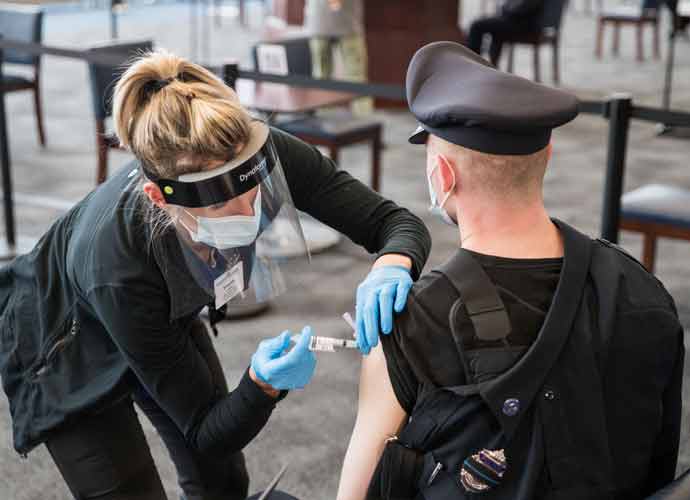
The Food and Drug Administration advisory committee unanimously voted 21-0 on Tuesday to recommend Novavax’s Covid-19 vaccination for emergency use in adults after a day-long meeting where they discussed data on safety and effectiveness.
The FDA will likely follow the committee’s suggestion, in signing off on Novavax’s two doses. If they do, pharmacies and health care providers will be able to begin administering doses for the fourth FDA-approved vaccine in the U.S.
The vaccine was developed differently than the other available vaccines. The technology in Novavax’s shots is more closely related to proven technology long used to create vaccines that fight hepatitis B and HPV. Pfizer and Moderna relied on using messenger RNA technology to protect against Covid-19. This could appeal to the millions of unvaccinated U.S. adults, some of which may have been opposed to using messenger RNA to be vaccinated.
Low rates of myocarditis (0.007%) and placebo (0.005%) were found in participants in the testing of the shots, mirroring participant reactions to the Pfizer/BioNTech and Moderna doses.
Subscribe to our free weekly newsletter!
A week of political news in your in-box.
We find the news you need to know, so you don't have to.
The U.S. would join multiple European countries, Australia, Canada, New Zealand, India, South Korea and more in authorizing the use of the Novavax doses.
Studies conducted in both the U.S. and Mexico, notably before the Omicron surge, proved the vaccine to be 90% effective protection from Covid-19 and completely effective in preventing serious illness.
Get the most-revealing celebrity conversations with the uInterview podcast!








Leave a comment